|
Iron forms compounds in a wide range of oxidation states, −4 to +7. 
Iron shares many properties of other transition metals, including the other group 8 elements, ruthenium and osmium. Ĭhemically, the most common oxidation states of iron are iron(II) and iron(III). 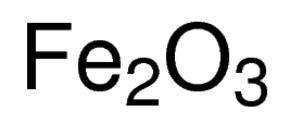
Iron is also the metal at the active site of many important redox enzymes dealing with cellular respiration and oxidation and reduction in plants and animals. To maintain the necessary levels, human iron metabolism requires a minimum of iron in the diet. These two proteins play essential roles in vertebrate metabolism, respectively oxygen transport by blood and oxygen storage in muscles. The body of an adult human contains about 4 grams (0.005% body weight) of iron, mostly in hemoglobin and myoglobin. electrolytic iron) are more resistant to corrosion. Unlike the oxides of some other metals that form passivating layers, rust occupies more volume than the metal and thus flakes off, exposing more fresh surfaces for corrosion. Iron reacts readily with oxygen and water to produce brown-to-black hydrated iron oxides, commonly known as rust. Pristine and smooth pure iron surfaces are a mirror-like silvery-gray. The iron and steel industry is thus very important economically, and iron is the cheapest metal, with a price of a few dollars per kilogram or pound. In the modern world, iron alloys, such as steel, stainless steel, cast iron and special steels, are by far the most common industrial metals, due to their mechanical properties and low cost. That event is considered the transition from the Bronze Age to the Iron Age. Humans started to master that process in Eurasia during the 2nd millennium BCE and the use of iron tools and weapons began to displace copper alloys-in some regions, only around 1200 BCE. It is the fourth most common element in the Earth's crust, being mainly deposited by meteorites in its metallic state, with its ores also being found there.Įxtracting usable metal from iron ores requires kilns or furnaces capable of reaching 1,500 ☌ (2,730 ☏) or higher, about 500 ☌ (932 ☏) higher than that required to smelt copper. It is, by mass, the most common element on Earth, just ahead of oxygen (32.1% and 30.1%, respectively), forming much of Earth's outer and inner core. It is a metal that belongs to the first transition series and group 8 of the periodic table. The degradation of cysteine releases hydrogen sulfide gas that reacts with the ferric citrate to produce ferrous sulfide.Iron is a chemical element with the symbol Fe (from Latin ferrum 'iron') and atomic number 26. Peptone iron agar contains the amino acid cysteine and a chemical indicator, ferric citrate. The presence of ferrous sulfide as a visible black precipitate in the growth medium peptone iron agar can be used to distinguish between microorganisms that produce the cysteine metabolizing enzyme cysteine desulfhydrase and those that do not. This reaction occurs more rapidly in older eggs as the whites are more alkaline. This color change is due to iron(II) sulfide, which forms as iron from the yolk reacts with hydrogen sulfide released from the egg white by the heat. When eggs are cooked for a long time, the yolk's surface may turn green. Pyrrhotite is a waste product of the Desulfovibrio bacteria, a sulfate reducing bacteria. These metal sulfides, such as iron(II) sulfide, are often black or brown, leading to the color of sludge. Some of the hydrogen sulfide will react with metal ions in the water or solid to produce iron or metal sulfides, which are not water-soluble. Iron sulfides occur widely in nature in the form of iron–sulfur proteins.Īs organic matter decays under low-oxygen (or hypoxic) conditions such as in swamps or dead zones of lakes and oceans, sulfate-reducing bacteria reduce various sulfates present in the water, producing hydrogen sulfide. In moist air, iron sulfides oxidize to hydrated ferrous sulfate. Iron sulfide reacts with hydrochloric acid, releasing hydrogen sulfide: FeS + 2 HCl → FeCl 2 + H 2S FeS + H 2SO 4 → FeSO 4 + H 2S 
All are black, water-insoluble solids.įeS can be obtained by the heating of iron and sulfur: Fe + S → FeSįeS adopts the nickel arsenide structure, featuring octahedral Fe centers and trigonal prismatic sulfide sites. Iron sulfides are often iron-deficient non-stoichiometric. sulphide) is one of a family chemical compounds and minerals with the approximate formula Fe S. Iron(II) sulfide or ferrous sulfide (Br.E.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
